Category Archives: Medical Device
Medical Device License Amendments in Thailand
Medical Device License Amendments in Thailand: A Complete Guide by Device Class Registered a medical... [read more]
May
Hong Kong Medical Device & IVD Registration | MDACS Overview
Medical Device and IVD Registration in Hong Kong: Regulatory Overview and Practical Considerations Hong Kong... [read more]
Jan
How to Secure a Medical Device License in Indonesia
How to Secure a Medical Device License in Indonesia — and Use Thailand as a... [read more]
Oct
Medical Devices in Thailand: Interview with Diego Sala at DIA Suzhou 2025
The 2025 DIA Suzhou International Innovative Medical Devices Exhibition brought together top industry professionals, regulators,... [read more]
Sep
Medical Device Submission in Thai FDA: Our Recent Work in Suzhou
Medical Device Submission in Thai FDA: Our Recent Work in Suzhou, to support Medical Device... [read more]
Aug
Medical Device Regulation in China: A Complete Guide
A comparison of Medical Device Regulation between China and Thailand Part 1: Overview of the... [read more]
Jul
Knowledge Empowers Business – Forum 2025: A Landmark Workshop on Food Supplements and Medical Devices
Knowledge Empowers Business – Forum 2025: A Landmark Workshop on Food Supplements and Medical Devices... [read more]
Jul
“Family of Medical Device” Concept in Thailand: What Variants Are Allowed?
When registering a family of medical device in Thailand, manufacturers often seek to reduce regulatory... [read more]
Jun
Medical Device License Transfer in Thailand: When and How It Can Be Done
Medical Device License Transfer in Thailand: When and How It Can Be Done In the... [read more]
Oct
Thai FDA IVD Risk Classification – IVD registration in Thailand
At Siam Trade Development, we pride ourselves on being one of the leading regulatory firms... [read more]
Oct
The New Abridged Channel for Medical Device Submission in Thailand
In a move to streamline the medical device registration process, the Thai FDA has introduced... [read more]
Jul
TFDA Medical Device GDP: Thailand issues new regulation for Good Distribution Practices
TFDA Medical Device GDP: Good Distribution Practices for Importers of Medical Devices in Thailand –... [read more]
Jun
Essential Licenses and Quality Certifications for Establishing a Medical Device Factory
Effective from July 3rd, 2024, to open a medical device factory according to the text... [read more]
Jun
Thai FDA Concise Program for Medical Device Registration
Concise evaluation and Reliance program 2024 is the notification ruling the Thai FDA Concise Program... [read more]
Apr
Thai FDA Medical Device: Understanding the Impact of Partial 2 Submission Termination
Thai FDA Medical Device process has changed. Thai FDA’s recent decision to terminate the medical... [read more]
Jan
Streamlining Medical Device Registration in Thai FDA: A Comprehensive Guide
Introducing Medical Device Registration in Thai FDA Navigating the regulatory landscape for medical device registration... [read more]
Dec
Registering injectable products as cosmetics: is it legal?
Registering injectable products as cosmetics: is it legal? On September 22, 2023, FDA published an... [read more]
Sep
Veterinary Medical Devices for Animals
Veterinary Medical Devices for Animals is a new niche going to develop quickly, considering that... [read more]
Aug
Timing for Thai FDA Medical Device Registration
Thai FDA amended their timing for Medical Device Registration, by shortening the process in some... [read more]
May
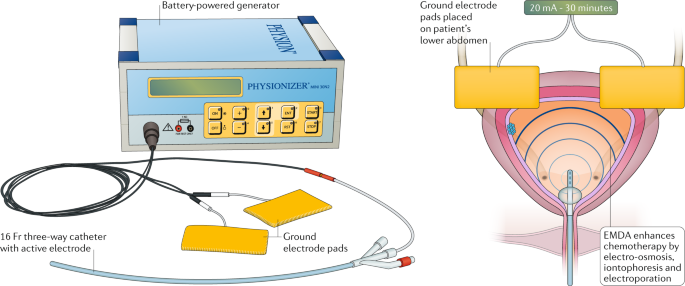
Electromotive Drug Administration EMDA – Mitomycin in bladder cancer treatment: a prospective observational study
A prospective observational study on Electromotive Drug Administration (EMDA®- Mitomycin) in bladder cancer treatment is... [read more]
Sep
THAI FDA Medical Device Classification – Risk based
Thai FDA Medical Device Classification – Which class is my Medical Device in Thailand? ... [read more]
May