Category Archives: Regulatory and Compliance
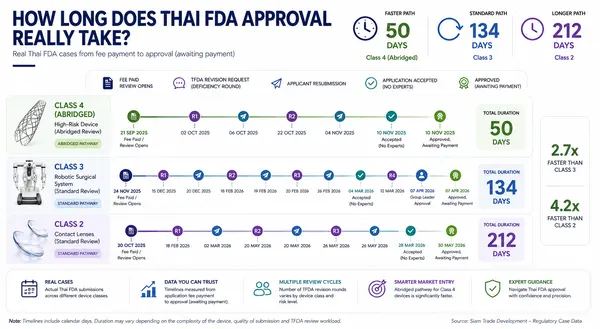
Thailand FDA Medical Device Registration Timeline
Thailand FDA Medical Device Registration Timeline: What the Data Actually Shows There is a question... [read more]
May
Medical Device License Amendments in Thailand
Medical Device License Amendments in Thailand: A Complete Guide by Device Class Registered a medical... [read more]
May
Thai FDA Registration for Chinese Manufacturers
How Chinese Manufacturers Enter the Thai FDA Market Thailand is a key destination for... [read more]
Apr
Shilajit Thailand: Legal Status, Thai FDA & Market Reality
Shilajit in Thailand: Is It Legal? Thai FDA Status, Risks, and Market Reality In recent... [read more]
Apr
Thai FDA Food Registration New Regulation 2026: Lab Testing After Submission
Thai FDA Food Registration New Regulation 2026: What Changes for Functional Food and Nutraceuticals The... [read more]
Mar
Thai FDA Nutrient Tolerance Criteria for Supplements
Thai FDA nutrient tolerance supplement in Dietary Supplement Labeling In February 2026, the Food Division... [read more]
Mar
Vitamins and minerals in dietary supplements: new Thai FDA guidance
Vitamins and minerals in dietary supplements: what really changes under the new Thai FDA guidance... [read more]
Feb
Shaping the Future of Foreign Business in Thailand: Regulatory Scenario and FBL Outlook
Foreign Business License Thailand: Regulatory Direction, Operational Trends and Implications for Foreign Investors Siam Trade... [read more]
Feb
Hong Kong Medical Device & IVD Registration | MDACS Overview
Medical Device and IVD Registration in Hong Kong: Regulatory Overview and Practical Considerations Hong Kong... [read more]
Jan
Functional Foods in Thailand: Regulatory Strategy Beyond Compliance
Functional Foods in Thailand | Regulatory Strategy, Claims & Thai FDA Framework Functional foods are... [read more]
Jan
Thai FDA Health Claims for Dietary Supplements: Notification 447 Explained
Thai FDA Health Claims for Dietary Supplements: A Practical Guide to Notification No. 447 Health... [read more]
Jan
Functional Food in Thailand: Siam Trade Development at FFWS 2026
Functional Food in Thailand: Siam Trade Development Invited as Regulatory Speaker at FFWS 2026 Siam... [read more]
Jan
The Hidden Risk Behind Renting a Warehouse for Medical Devices in Thailand
The Hidden Risk Behind Renting a Warehouse for Medical Devices in Thailand: A Legal Loophole... [read more]
Dec
Thai FDA submission process: Summary of Verification and Validation
The Thai FDA submission process for medical devices requires complete and precise documentation, and the... [read more]
Sep
Medical Devices in Thailand: Interview with Diego Sala at DIA Suzhou 2025
The 2025 DIA Suzhou International Innovative Medical Devices Exhibition brought together top industry professionals, regulators,... [read more]
Sep
The Myth of Sole Proprietorship and Single Shareholder Limited Companies in Thailand: Legal Realities Unveiled
In 2021, we dispelled one of the common myths circulating among investors and even some... [read more]
Aug
Medical Device Regulation in China: A Complete Guide
A comparison of Medical Device Regulation between China and Thailand Part 1: Overview of the... [read more]
Jul
Knowledge Empowers Business – Forum 2025: A Landmark Workshop on Food Supplements and Medical Devices
Knowledge Empowers Business – Forum 2025: A Landmark Workshop on Food Supplements and Medical Devices... [read more]
Jul
Cannabis Announcement 23 June 2025: What It Really Means
On June 23, 2025, the Thai Ministry of Public Health issued a new Cannabis Announcement... [read more]
Jun
“Family of Medical Device” Concept in Thailand: What Variants Are Allowed?
When registering a family of medical device in Thailand, manufacturers often seek to reduce regulatory... [read more]
Jun
Cosmetic Advertising License in Thailand
Cosmetic Advertising License in Thailand – Summary Guide When a Cosmetic Advertising License is Required Technically,... [read more]
Apr
Singapore Market Immersion Program for Projects in Thailand
The Singapore Market Immersion Program (MIP) is a strategic initiative designed to assist Singaporean enterprises... [read more]
Mar
Thai FDA Announces New Forbidden Cosmetic Ingredients
Thai FDA Published the list of Forbidden Cosmetic Ingredients The Thai Food and Drug Administration... [read more]
Feb
Mule Accounts in Thailand: better the problem or the solution?
The Thai Government’s Crackdown on Mule Accounts: A Sensible Solution or a New Problem? The... [read more]
Feb